TEST Sponsor and Provider: Two Very Different Perspectives on Risk-based Provider Oversight

The advent of ICH E6 (R2), with its risk-based approaches to provider oversight, has not been one of the smoother transitions in the industry. The Avoca Group’s research, documented in the 2017 Avoca Industry Report, illuminates significant concerns about this addendum by sponsors and providers alike. These concerns, however, serve to point the way toward a more efficient and effective method of managing risk.
Is your company prepared?
The story begins with the simple question, “Is your company prepared to address changes to oversight resulting from ICH E6 (R2)?”
Almost three-quarters of providers (74%) agree or strongly agree that their company is prepared for these changes. That is understandable, since contract research organizations (CROs) have always had the responsibility to oversee any subcontractors they bring on. Plus, it should be noted that three-quarters of the provider survey respondents were CROs.
Sponsors, however, are not as confident, with only 58% indicating agreement/strong agreement. Sponsors are likely feeling pressure as they are now faced not only with overseeing the CRO they hire, but also any subcontractors the CRO utilizes.
There is a definite risk associated with having ultimate responsibility for a process while being one or two steps removed from it, if sufficient quality oversight is not present.
How are oversight practices working out?
Oversight practices can be a point of contention between sponsors and providers. Avoca asked sponsors to assess themselves in the areas of documentation and roles, and then asked providers to assess sponsors in the same areas.
As the chart below shows, the majority of sponsors feel fairly positive about their approach to documentation and roles/responsibilities. Most providers, however, don’t think sponsors are doing a great job.
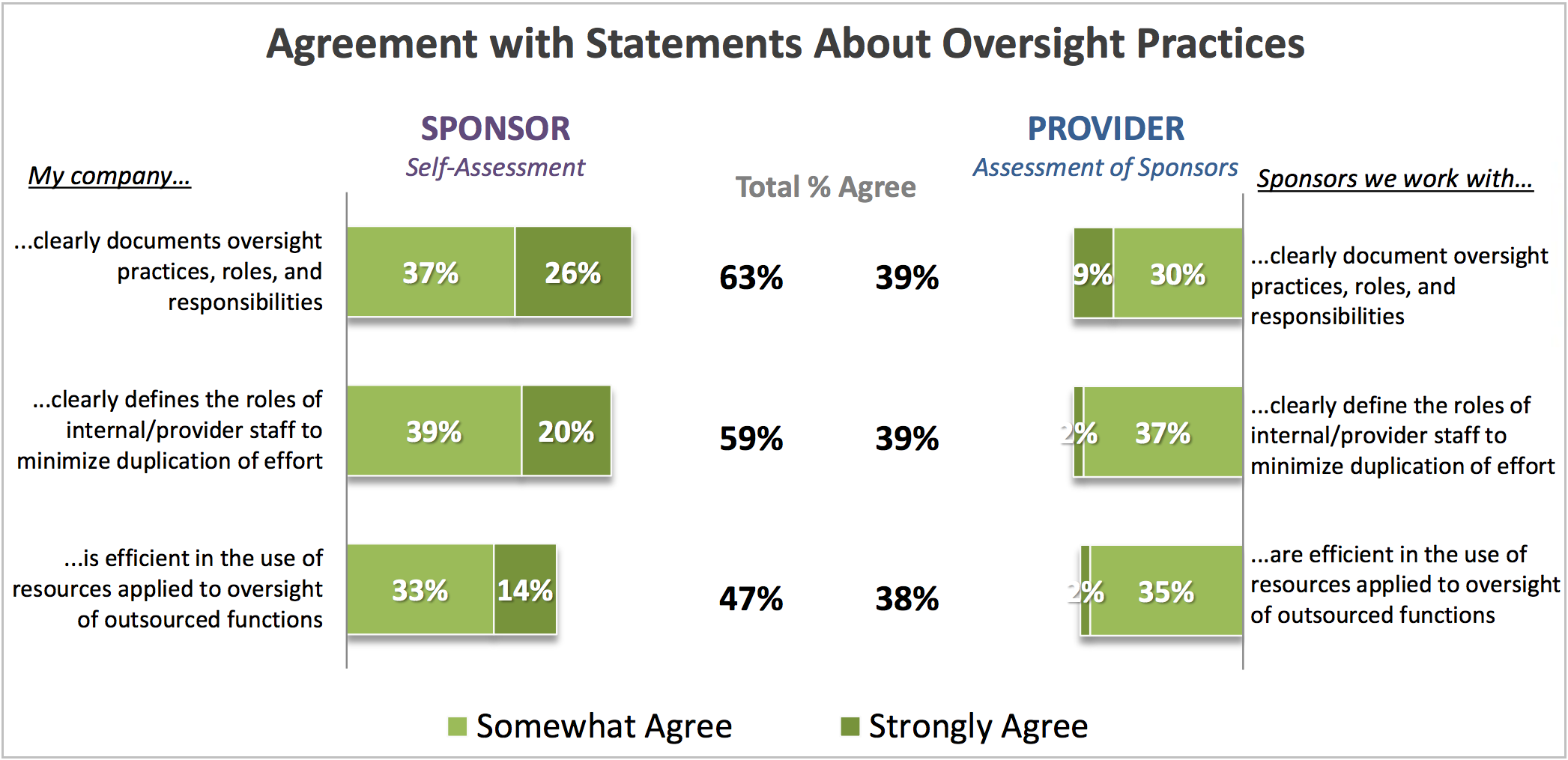
What is the basis of this discrepancy in perspective? First, consider documentation. Providers rely upon documentation to do their job and fulfill sponsor expectations; they have a vested interest in that documentation being detailed and precise. Sponsors, on the other hand, may view documentation from a “defensive” position. That is, sponsors may prefer a high-level approach to documentation that allows greater interpretive freedom. This approach gives sponsors greater confidence in their ability to demonstrate compliance, but it can be at the expense of the specificity providers’ desire.
The next point of discrepancy involves defining roles to minimize duplication of effort. As with documentation, there appear to be divergent views between sponsors and providers. The third question shown in the chart, which asks about efficient use of resources, points to a likely reason for this discrepancy: namely, relatively low levels of agreement from both sponsor and provider perspectives with respect to the efficient use of resources suggests that, even if roles are clearly defined, work is still being duplicated on the job. Because sponsors have been given oversight responsibility, they may feel it incumbent upon them to duplicate work to verify outcomes. Providers, of course, view this as unnecessary and indicative of a lack of trust on the part of the sponsor.
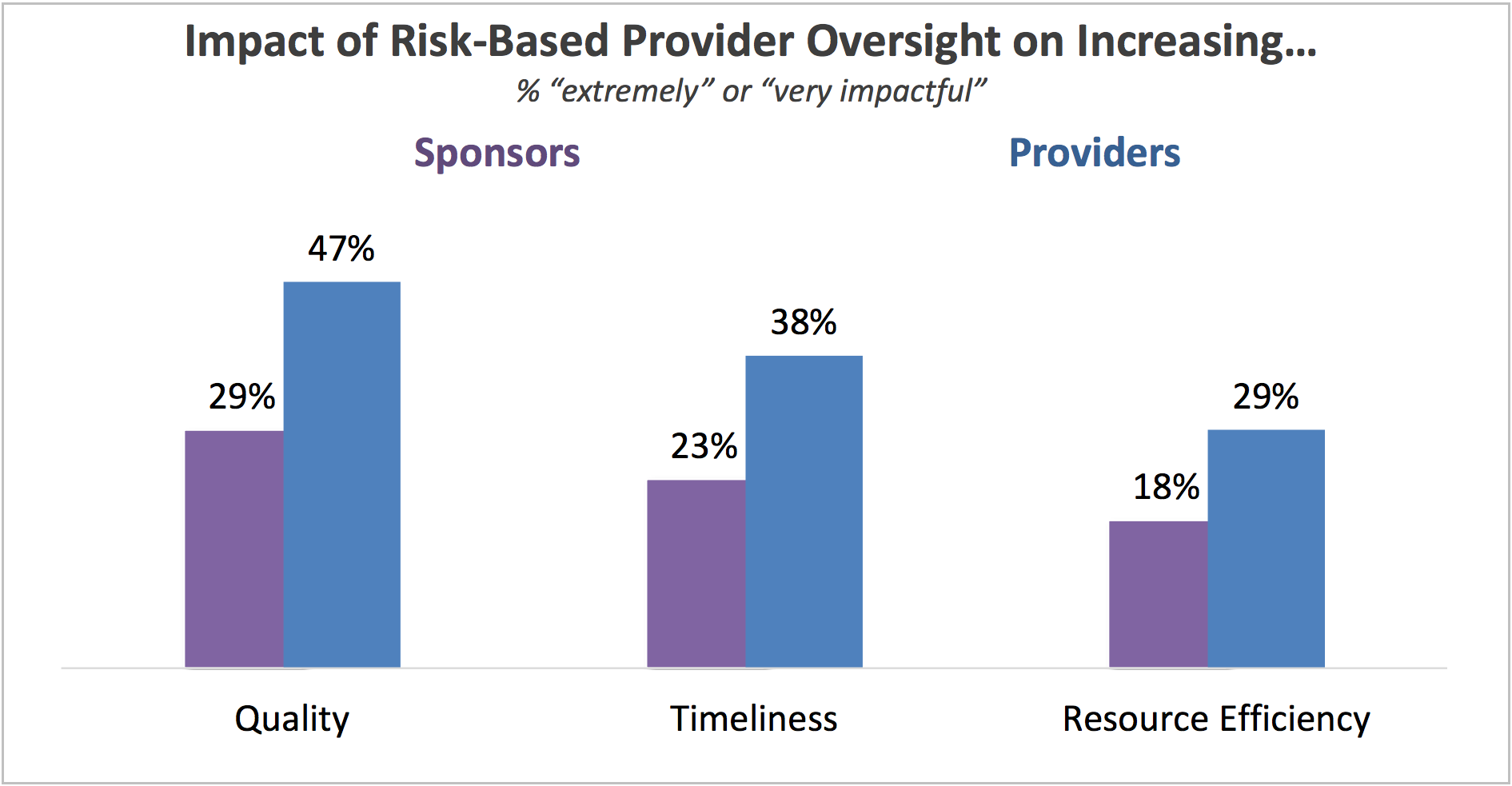
How is risk-based provider oversight working out?
All that being said, what do sponsors and providers think about the impact of using risk-based provider oversight in terms of increasing quality, timeliness, and resource efficiency? The answer from both parties: not much.
How do we make this work – together?
For sponsors and providers to realize sustainable changes oversight practices as allowed by ICH E6 (R2), risk-based approaches must deliver quantitative benefits for quality, timeliness, and resource efficiency – far more so than it is currently doing. The data shown above from the 2017 Avoca Industry Report highlights three areas where sponsors and providers can improve to gain those benefits:
- Providers can share best practices with sponsors to help sponsors become more comfortable with their new oversight role.
- Sponsors and providers must define expectations for documentation and come to mutual agreement regarding its purpose, scope, and level of detail.
- Sponsors must establish a level of trust with providers to eliminate duplication of effort.
Risk-based provider oversight is an area of tremendous opportunity for sponsor and provider alike. To gain the value that it promises, however, sponsors and providers will need to work in closer collaboration and partnership than ever before.
Download Avoca’s 2017 Provider Oversight Industry Report.
Author:
 |
Dennis Salotti
Vice President, Operations, The Avoca Group |

